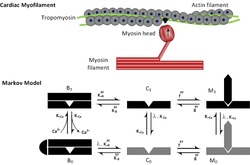
- Markov Model of Cardiac Thin Filament Ca 2+ Independent Activation
Conceptual and computational models have generally assumed that each regulatory unit (RU) of the thin filament remains in the blocked state until Ca 2+ binds to troponin C. Previous models, failed to fully resolve situations in which some fraction of myofilament troponin C is replaced with a non-Ca 2+ binding mutant (xTnC). This effect was caused by the assumption of strong cooperative inhibition among nearest-neighbor RUs, which was required to produce basic myofilament activation behavior. We hypothesized that permitting some Ca 2+ - independent RU activation while maintaining cooperative inhibition would reconcile this discrepancy. Our Markov Chain Monte Carlo (MCMC) simulations suggest that thin filament function does not require perfect Ca 2+ switch fidelity.
Conceptual and computational models have generally assumed that each regulatory unit (RU) of the thin filament remains in the blocked state until Ca 2+ binds to troponin C. Previous models, failed to fully resolve situations in which some fraction of myofilament troponin C is replaced with a non-Ca 2+ binding mutant (xTnC). This effect was caused by the assumption of strong cooperative inhibition among nearest-neighbor RUs, which was required to produce basic myofilament activation behavior. We hypothesized that permitting some Ca 2+ - independent RU activation while maintaining cooperative inhibition would reconcile this discrepancy. Our Markov Chain Monte Carlo (MCMC) simulations suggest that thin filament function does not require perfect Ca 2+ switch fidelity.

- Topological Simulated Orientation Atlas of the Cardiac Muscle Fibers
Topological organization of heart muscle fibers and their orientation play an important role in the blood pumping function in an intact heart. Contractility dysfunction for instance often raise from pathologies that affect the myocyte cells. Therefore it is essential to adequately model the cardiac fibers and their variations across the heart wall to get reliable finite element simulations. In this project, we used a coupled electromechanical FEM model to simulate a human truncated bi-ventricular 3-D geometries .
Topological organization of heart muscle fibers and their orientation play an important role in the blood pumping function in an intact heart. Contractility dysfunction for instance often raise from pathologies that affect the myocyte cells. Therefore it is essential to adequately model the cardiac fibers and their variations across the heart wall to get reliable finite element simulations. In this project, we used a coupled electromechanical FEM model to simulate a human truncated bi-ventricular 3-D geometries .

- Mathematical Model of Aortic Valve Dynamics During Systole
A mathematical model that governs the ventricular-aortic valve dynamic and ejection blood flow transport during systole is derived in this article. The model presents a realistic coupling between aortic valve dynamics, sinus vortex local pressure, and variations in the systemic vascular resistance. The coupling is introduced by using the classical Hill's semi-spherical vortex model and the aortic pressure-area compliance constitutive relationships respectively. The effects induced by the existence of aortic sinus eddy vortices and using variable systemic vascular resistance on the overall valve opening-closing dynamics, as well as on the left ventricular and aortic pressures, blood flowrate, and aortic orifice area are examined. In additions, the strength of sinus vortex has coupled to the valve opening angle explicitly, and implicitly to the aortic orifice area in order to show the vortex strength development/decay during the four known opening-closing phases (fast-opening, fully-opening, slow-closing, and fast-closing) regimes.
A mathematical model that governs the ventricular-aortic valve dynamic and ejection blood flow transport during systole is derived in this article. The model presents a realistic coupling between aortic valve dynamics, sinus vortex local pressure, and variations in the systemic vascular resistance. The coupling is introduced by using the classical Hill's semi-spherical vortex model and the aortic pressure-area compliance constitutive relationships respectively. The effects induced by the existence of aortic sinus eddy vortices and using variable systemic vascular resistance on the overall valve opening-closing dynamics, as well as on the left ventricular and aortic pressures, blood flowrate, and aortic orifice area are examined. In additions, the strength of sinus vortex has coupled to the valve opening angle explicitly, and implicitly to the aortic orifice area in order to show the vortex strength development/decay during the four known opening-closing phases (fast-opening, fully-opening, slow-closing, and fast-closing) regimes.
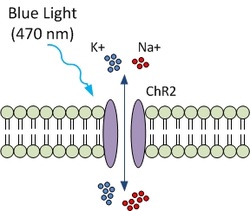
- Computational Optogenetics:
Optogenetic approaches allow cellular membrane potentials to be perturbed by light.
When applied to muscle cells, mechanical events can be controlled through a process
that could be termed (optomechanics.) Besides functioning as an optical on/off switch,
we hypothesized that optomechanical control could include the ability to manipulate the
strength and duration of contraction events. To explore this possibility, we constructed
an electromechanical model of the human ventricular cardiomyocyte while adding a representation
of channelrhodopsin-2 (ChR2), a light-activated channel commonly used in optogenetics.
Two hybrid stimulus protocols were developed that combined light-based
stimuli with traditional electrical current (all-or-none) excitation. The first protocol
involved delivery of a subthreshold optical stimulus followed 50–90 ms later by an electrical
stimulus. The result was a graded inhibition of peak cellular twitch force in concert
with a prolongation of the intracellular Ca-transient. The second protocol was comprised
of an electrical stimulus followed by a long light pulse (250–350 ms) that acted to
prolong the cardiac action potential (AP).
Optogenetic approaches allow cellular membrane potentials to be perturbed by light.
When applied to muscle cells, mechanical events can be controlled through a process
that could be termed (optomechanics.) Besides functioning as an optical on/off switch,
we hypothesized that optomechanical control could include the ability to manipulate the
strength and duration of contraction events. To explore this possibility, we constructed
an electromechanical model of the human ventricular cardiomyocyte while adding a representation
of channelrhodopsin-2 (ChR2), a light-activated channel commonly used in optogenetics.
Two hybrid stimulus protocols were developed that combined light-based
stimuli with traditional electrical current (all-or-none) excitation. The first protocol
involved delivery of a subthreshold optical stimulus followed 50–90 ms later by an electrical
stimulus. The result was a graded inhibition of peak cellular twitch force in concert
with a prolongation of the intracellular Ca-transient. The second protocol was comprised
of an electrical stimulus followed by a long light pulse (250–350 ms) that acted to
prolong the cardiac action potential (AP).
